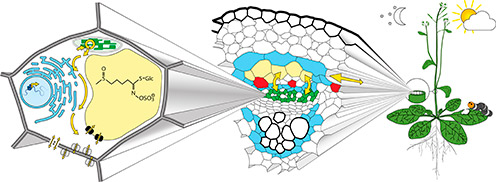
Defense compounds on the move
According to the optimal defense theory, plants accumulate defense compounds to the highest level in those tissues with the highest fitness value while other tissues are often the site of production. Thus, we know that defense compounds are mobile. Moreover, due to their high metabolic cost plants appear to reuse defense compounds by remobilizing and transporting them to new storage tissues. An important prerequisite for studying mobility of a given metabolite is to know both where the metabolite is de novo biosynthesized and where it is stored at the cellular and subcellular level.
What are the genes and processes involved in transport of defense compounds? How are they controlled to deliver the right amount of defense compounds to the correct location at the correct time during plant development? We address these questions using the glucosinolate defense compounds,in the model plant Arabidopsis. We know that glucosinolates first accumulate in green leaves and roots, then in reproductive organs such as flowers and lastly in seeds. In addition, Arabidopsis root exudes glucosinolates to the rhizosphere. Thus, we know that glucosinolates are very mobile.
Towards our goal of understanding glucosinolate mobility from source to sink our first aim is to identify the specific cell types involved. Evidence suggests that glucosinolates are produced in specific cell types embedded in the vasculature and stored in laticifer-like S-cells along the vasculature and the leaf margin. Currently, we are identifying glucosinolate-producing cells in Arabidopsis plants using transgenic lines with fluorophore-tagged biosynthetic enzymes and characterizing these cells with correlative transmission electron microscopy.
At the subcellular level, we are investigating how the glucosinolates are translocated from the ER-associated production site to storage in the S-cells. Does short distance glucosinolate transport from cell to cell include transporter proteins or plasmodesmata?
Ultimately, we want to unravel the mechanism behind S-cell accumulation, root exudation and seed loading using single cell metabolite analyses, TEM, grafting and bioimaging approaches, single seed analyses from segregating populations, and root exudation assays.
Christa Kanstrup, and Hussam Hassan Nour-Eldin. "The emerging role of the nitrate and peptide transporter family, NPF in plant specialized metabolism" Current opinion in plant biology (2022): 102243. DOI:10.1016/j.pbi.2022.102243
Kazachkova Y, Zemach I, Panda S, Bocobza S, Vainer A, Rogachev I, Dong Y, Ben-Dor S, Veres D, Kanstrup C, Lambertz SK, Crocoll C, Hu Y, Shani E, Michaeli S, Nour-Eldin, Hussam Hassan, Zamir D, Aharoni A (2021) The GORKY glycoalkaloid transporter is indispensable for preventing tomato bitterness. Nature Plants 7, no. 4 (2021): 468-480. DOI:10.1038/s41477-021-00865-6
Yang, Zhi-Ling, Nour-Eldin, Hussam Hassan, Sabine Hänniger, Michael Reichelt, Christoph Crocoll, Fabian Seitz, Heiko Vogel, and Franziska Beran. "Sugar transporters enable a leaf beetle to accumulate plant defense compounds." Nature communications 12, no. 1 (2021): 1-12. DOI:10.1038/s41467-021-22982-8
Xu D, Hunziker P, Koroleva O, Blennow A, Crocoll C, Schulz A, Nour-Eldin HH, Halkier BA (2019) GTR-mediated radial import directs accumulation of defensive glucosinolates to sulfur-rich cells (S-cells) in phloem cap of inflorescence stem of Arabidopsis thaliana. Mol Plant. DOI: 10.1016/j.molp.2019.06.008
Hunziker P, Halkier BA, Schulz A (2019) Arabidopsis glucosinolate storage cells transform into phloem fibres at late stages of development. J Exp Bot. DOI:10.1093/jxb/erz176
Nour-Eldin HH, Madsen SR, Engelen S, Jørgensen ME, Olsen CE, Andersen JS, Seynnaeve D, Verhoye T, Fulawka R, Denolf P, Halkier BA (2017) Reduction of antinutritional glucosinolates in Brassica oilseeds by mutation of genes encoding transporters. Nat Biotechnol. DOI: 10.1038/nbt.3823
Payne RME, Xu D, Foureau E, Teto Carqueijeiro MIS, Oudin A, Bernonville TD de, Novak V, Burow M, Olsen C-E, Jones DM, Tatsis EC, Pendle A, Halkier BA, Geu-Flores F, Courdavault V, Nour-Eldin HH, O’Connor SE (2017) An NPF transporter exports a central monoterpene indole alkaloid intermediate from the vacuole. Nature Plants 3: 16208. DOI: 10.1038/nplants.2016.208
Xu D, Hanschen FS, Witzel K, Nintemann SJ, Nour-Eldin HH, Schreiner M, Halkier BA (2016) Rhizosecretion of stele-synthesized glucosinolates and their catabolites requires GTR-mediated import in Arabidopsis. J Exp Bot. DOI: 10.1093/jxb/erw355
Darbani B, Motawia MS, Olsen CE, Nour-Eldin HH, Møller BL, Rook F (2016) The biosynthetic gene cluster for the cyanogenic glucoside dhurrin in Sorghum bicolor contains its co-expressed vacuolar MATE transporter. Sci Rep 6: 37079. DOI:10.1038/srep37079
Jørgensen ME, Nour-Eldin HH, Halkier BA (2015) Transport of defense compounds from source to sink: lessons learned from glucosinolates. Trends Plant Sci 20: 508–514. DOI: 10.1016/j.tplants.2015.04.006
Madsen SR, Olsen CE, Nour-Eldin HH, Halkier BA (2014) Elucidating the role of transport processes in leaf glucosinolate distribution. Plant Physiol 166: 1450–1462. DOI: 10.1104/pp.114.246249
Andersen TG, Nour-Eldin HH, Fuller VL, Olsen CE, Burow M, Halkier BA (2013) Integration of biosynthesis and long-distance transport establish organ-specific glucosinolate profiles in vegetative Arabidopsis. Plant Cell 25: 3133–3145. DOI: 10.1105/tpc.113.110890
Nour-Eldin HH, Andersen TG, Burow M, Madsen SR, Jørgensen ME, Olsen CE, Dreyer I, Hedrich R, Geiger D, Halkier BA (2012) NRT/PTR transporters are essential for translocation of glucosinolate defence compounds to seeds. Nature 488: 531–534. DOI: 10.1038/nature11285
Video
Video abstract of Jørgensen ME, Nour-Eldin HH and Halkier BA (2015) Transport of defense compounds from source to sink: lessons learned from glucosinolates. Trends in Plant Science
Contact persons
 |
Professor |
 |
Associate Professor Hussam H. Nour-Eldin huha@plen.ku.dk +45 353-698 |
 |
Professor Alexander Schulz als@plen.ku.dk +45 353-33350 |
 |
Assist. professor |
